Lithium chloride (LiCl) is used in a wide range of industrial processes, but it is best known as a highly effective agent for dehumidification and drying of process air. In industries where air conditions directly impact product quality, hygiene, and efficiency, LiCl is often a reliable choice.
In this blog, we guide you through the key topics: what LiCl is, why it works so well, where it comes from, how to apply it (with a focus on dehumidification), which concentration you need, what to consider regarding quality, how to work safely, and how to avoid pitfalls and misconceptions.
1. Basics: What is lithium chloride (LiCl) and where does it come from?
Lithium chloride is an inorganic salt composed of lithium (Li⁺) and chloride (Cl⁻). In its pure form, it is a white, crystalline material. The key property for industrial drying and dehumidification is that LiCl is strongly hygroscopic: it attracts water and can effectively bind water molecules.
That is why LiCl is often used in systems where you want to dehumidify process air without first having to cool it to extremely low temperatures.
Why does LiCl bind moisture so well?
At a molecular level, LiCl “holds” water because the ions (Li⁺ and Cl⁻) attract water molecules and stabilize them in solution. In practice, this means that when air comes into contact with an LiCl solution, water vapor can be absorbed relatively quickly into the liquid.
Where does lithium chloride come from?
The origin of LiCl can broadly be split into two parts:
- A) Where does the lithium come from?
Lithium is extracted from:
- Brines (salt lakes or underground brine sources): through evaporation and processing.
- Hard-rock minerals (such as spodumene): through mining and chemical processing.
The end product from this chain is often a lithium compound (e.g., lithium carbonate or lithium hydroxide), which is then further converted.
- B) How is LiCl produced?
Industrially, lithium chloride is typically produced by chemically converting a lithium source into the chloride form, for example by reaction with hydrochloric acid (HCl) or via other chloride-based routes. The exact production route differs by manufacturer, but the objective is the same: a solution or crystalline product with controlled purity.
Solid LiCl vs. 40% LiCl solution
In practice, many drying and dehumidification installations operate with an LiCl solution of around 40%. This is LiCl dissolved in water, at a concentration chosen to:
- provide sufficient moisture absorption capacity,
- be easy to pump and spray,
- remain stable in the process (without unwanted crystallization or excessive corrosion risk).
2. LiCl in dehumidification and drying: how does the process work?
LiCl systems work based on contact between humid air and a hygroscopic liquid.
- Dehumidification (absorption phase)
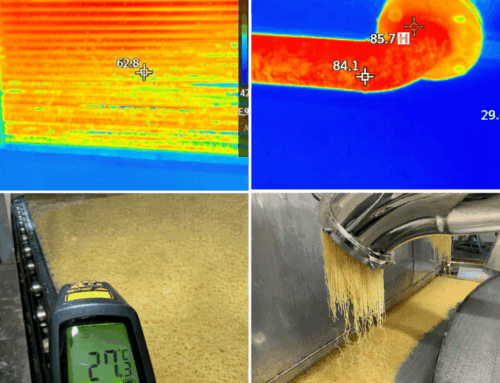
Humid process air flows through the dehumidifier. Through spray nozzles, packing, and contact surface area, the air comes into contact with the LiCl solution. The solution absorbs water vapor from the air. The air leaves the system drier, without having to be deeply cooled first. - Dilution and performance loss
The more water LiCl absorbs, the more the solution becomes diluted. As a result, its moisture-absorbing capacity decreases. - Regeneration (reconcentration)
The diluted LiCl solution is sent to the regenerator, where it is heated. The absorbed water evaporates and the solution becomes concentrated again. The restored LiCl solution then flows back to the dehumidification section.
In short: LiCl “captures” moisture from the air and is then “recharged” through regeneration.
3. Applications by industry: where is LiCl relevant?
LiCl is used worldwide in industries where process air and moisture control are critical:
- Meat industry (red and white meat)
Control of condensation, hygiene, a more stable process, and less product loss due to unwanted moisture load.
- Gelatin industry
A classic domain for air dehumidification and drying, with high demands for constant conditions.
- Pharmaceutical and fine chemicals
Where moisture can directly affect stability, powder behavior, and quality standards. - Food and ingredients
Think of processes where caking, sticking, or variation in drying is undesirable.
4. Why LiCl 40%?
A concentration such as 40% is a practical optimum in many installations.
- Concentration too low
Less moisture absorption → lower dehumidification capacity → risk of not achieving your target dew point/conditions. - Concentration too high
May lead to higher viscosity (harder to spray/pump), a higher risk of crystallization during local cooling/evaporation, and in some cases additional attention to material compatibility.
In practice, the “right” concentration depends on the system design and the required air conditions. That is why it is essential to measure and control within the engineering range.
5. Quality and purity
In the market, LiCl is sometimes offered under different labels. Key points to consider:
- Purity and impurities
Contamination can cause foaming, deposits, performance loss, or additional maintenance. - Certificate of Analysis (COA)
The certificate provides insight into composition/quality and ensures traceability. - Compatibility with different systems
Most LiCl dehumidification systems operate on similar principles, but solution quality and stability make a real difference in performance and maintenance.
In short: controlled supply and proven quality provide peace of mind in performance and maintenance.
6. Safety and handling: working safely with LiCl
LiCl solution requires professional handling:
- Use appropriate personal protective equipment as per the SDS/safety data sheet instructions.
- Maintain a tidy, dry workplace: spills can cause slipping hazards and corrosion issues in the surrounding area.
- Pay attention to materials. Material selection and condition (corrosion/crystals) play a role in and around LiCl systems.
- Follow proper procedures for storage, topping up, and sampling (to prevent contamination).
Tip: Make “cleanliness” a process step. Many practical issues originate from minor contamination or careless handling.
7. Myths and misconceptions about LiCl
Myth 1: “LiCl is always corrosive.”
Reality: LiCl can accelerate corrosion under certain conditions, but corrosion behavior strongly depends on material selection, concentration, temperature, maintenance condition, and impurities. It can be managed.
Myth 2: “The higher the concentration, the better.”
Reality: Above an optimum, side effects may occur (viscosity, spray behavior, crystallization risk). “Better” means staying within the design range.
Myth 3: “All LiCl is the same.”
Reality: Quality, impurities, and stability differ. This shows up in foaming, deposits, performance, and maintenance.
8. Logistics and supply: stock, packaging, and continuity
For a stable process, you always want sufficient LiCl available.
- Packaging formats
We supply standard drums of 50L, 200L, and 1000L IBCs, depending on your consumption, storage space, and handling. - Inventory management
Maintain the right stock levels: ordering “just too late” is often more expensive in moisture-critical processes than keeping an extra drum on hand. - Worldwide shipping and documentation
At TEMA Process, we are familiar with all required documentation and know what it takes to deliver lithium chloride to our customers anywhere in the world.
Finally
Lithium chloride is a powerful tool for industrial dehumidification because it binds moisture without requiring extremely deep cooling. Success is not only about the salt itself, but about the full picture: the right concentration, proven quality, safe handling, and a process you monitor regularly.
Need LiCl quickly? Request a no-obligation quotation for delivery in 50L or 200L drums. For larger volumes, we offer 1000L IBCs.